I prefer Soundhound better than shazam. Log in to Reply. Bobby Drake says: June 19, 2011 at 5:26 am. I used to be a Soundhound user, and my friend is a Shazam user. I say “used to be” because. Listen to Better Than by John Butler Trio, 127,694 Shazams, featuring on John Butler Trio Essentials, and Inspired by Powderfinger Apple Music playlists. Discovered using Shazam, the music discovery app. SoundHound: SoundHound is probably the best alternative to Shazam when it comes to music identification. No other app comes so close to Shazam. SoundHound identifies the song in an instant and also provides you with the lyrics to the song. A quick review of the DCUE's latest SHAZAM! From the early screenings. I give you my spoiler free thoughts right after seeing the film. Full spoiler review c. Midomi.com is a web-based tool provided by a SoundHound, a Shazam competitor. This is the closest thing there is a to a web-based version of Shazam. Or ask Cortana on windows 10. 

Valence Electrons: 4s 1 Electron Dot Model. Chemical Properties of Potassium. Electrochemical Equivalent: 1.4587g/amp-hr. Potassium - K (EnvironmentalChemistry.com)- Comprehensive information for the element Potassium - K is provided by this page including scores of properties, element names in many languages, most known nuclides. Question: Atom #1 Number Of Valence Electrons Prediction Of Charge Atom #2 Of Valence Electrons Prediction Molecular Of Charge Formula Name Of Compe Na 11 8 Na2O Sodium Oxide +1 KF Potassium Fluoride Mg +2 MgCl2 Magnesium Chloride +2 IN Ca3N2 Calcium Nitride ΑΙ +3 A1253 Aluminum Sulfide Part 2: Covalent Bonding 1. You Will First Investigate 5 Diatomic Molecules. A step-by-step explanation of how to draw the KBr Lewis Dot Structure.For KBr we have an ionic compound and we need to take that into account when we draw th. Valence electrons are generally regarded as being 'the outermost electrons' for a given atom. Therefore, with neutral potassium, there is one valence electron. If we take away the outermost electron, we now have a new set of outermost electrons being the 8 electrons in the 3s and 3p orbitals. Ron is correct in his assessment of semantics. The valence electrons in the potassium ions present in potassium nitrate are a. The 2p electrons only b. The 3s electrons only c. The 3s and 3p electrons only d. The 3p electrons only e. There are no valence electrons in the potassium ions in the compound above 2. Which one of the following reactions is exothermic to a substantial degree?
Element Potassium - K
Comprehensive data on the chemical element Potassium is provided on this page; including scores of properties, element names in many languages, most known nuclides of Potassium. Common chemical compounds are also provided for many elements. In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies.
Potassium Menu
- Potassium Page One
- Potassium Page Two
- Potassium Page Three
Overview of Potassium
Potassium Valence Electrons
- Atomic Number: 19
- Group: 1
- Period: 4
- Series: Alkali Metals
Potassium's Name in Other Languages
- Latin: Kalium
- Czech: Draslík
- Croatian: Kalij
- French: Potassium
- German: Kalium - s
- Italian: Potassio
- Norwegian: Kalium
- Portuguese: Potássio
- Russian: Калий
- Spanish: Potasio
- Swedish: Kalium
Atomic Structure of Potassium
- Atomic Radius: 2.77Å
- Atomic Volume: 45.46cm3/mol
- Covalent Radius: 2.03Å
- Cross Section (Thermal Neutron Capture) σa/barns: 2.1
- Crystal Structure: Cubic body centered
- Electron Configuration:
- 1s2 2s2p6 3s2p6 4s1
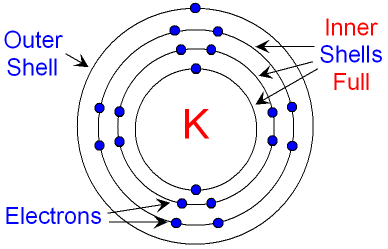
- Electrons per Energy Level: 2,8,8,1
- Shell Model
- Shell Model
- Ionic Radius: 1.38Å
- Filling Orbital: 4s1
- Number of Electrons (with no charge): 19
- Number of Neutrons (most common/stable nuclide): 20
- Number of Protons: 19
- Oxidation States: 1
- Valence Electrons: 4s1
- Electron Dot Model
- Electron Dot Model
Chemical Properties of Potassium
Potassium Chloride Valence Electrons
- Electrochemical Equivalent: 1.4587g/amp-hr
- Electron Work Function: 2.3eV
- Electronegativity: 0.82 (Pauling); 0.91 (Allrod Rochow)
- Heat of Fusion: 2.334kJ/mol
- Incompatibilities:
- Ionization Potential
- First: 4.341
- Second: 31.625
- Third: 45.72
- Valence Electron Potential (-eV): 10.4
Physical Properties of Potassium
- Atomic Mass Average: 39.0983
- Boiling Point: 1032K 759°C 1398°F
- Coefficient of lineal thermal expansion/K-1: 83E-6
- Conductivity
- Electrical: 0.139 106/cm Ω
Thermal: 1.024 W/cmK
- Electrical: 0.139 106/cm Ω
- Density: 0.862g/cc @ 300K
- Description:
- Very soft silvery metal that quickly oxidizes to white in air and reacts violently in water.
- Elastic Modulus:
- Bulk: 3.1/GPa
- Rigidity: 1.3/GPa
- Youngs: 3.53/GPa
- Enthalpy of Atomization: 89.5 kJ/mole @ 25°C
- Enthalpy of Fusion: 2.32 kJ/mole
- Enthalpy of Vaporization: 77.5 kJ/mole
- Flammablity Class:
- Freezing Point:see melting point
- Hardness Scale
- Brinell: 0.363 MN m-2
- Mohs: 0.4
- Heat of Vaporization: 79.87kJ/mol
- Melting Point: 336.5K 63.35°C 146.03°F
- Molar Volume: 45.46 cm3/mole
- Physical State (at 20°C & 1atm): Solid
- Specific Heat: 0.75J/gK
- Vapor Pressure = [email protected]°C
Regulatory / Health
- CAS Number
- 7440-09-7
- RTECS: TS6465000
- OSHAPermissible Exposure Limit (PEL)
- No limits set by OSHA
- OSHA PEL Vacated 1989
- No limits set by OSHA
- NIOSHRecommended Exposure Limit (REL)
- No limits set by NIOSH
- Levels In Humans:
Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances.- Blood/mg dm-3: 1620
- Bone/p.p.m: 2100
- Liver/p.p.m: 16000
- Muscle/p.p.m: 16000
- Daily Dietary Intake: 1400-7400 mg
- Total Mass In Avg. 70kg human: 140 g

Who / Where / When / How
- Discoverer: Sir Humphrey Davy
- Discovery Location: London England
- Discovery Year: 1807
- Name Origin:
- From potash (pot ash); K from latin: kalium
- Abundance of Potassium:
- Earth's Crust/p.p.m.: 21000
- Seawater/p.p.m.: N/A
- Atmosphere/p.p.m.: N/A
- Sun (Relative to H=1E12): 145000
- Sources of Potassium:
- Found in minerals like carnallite [(KMgCl3).6H2O], sylvite (KCL) and alunite. Annual world production is around 200 tons. Primary mining areas are in Germany, Spain, Canada, USA and Italy.
- Uses of Potassium:
- Used as potash in making glass, soap, lenses and salt substitute. Also as saltpeter, potassium nitrate (KNO3) it is used to make explosives and to color fireworks in mauve.
- Additional Notes:
- Formerly called kalium hence the symbol 'K'.
Potassium Menu
- Potassium Page One
- Potassium Page Two
- Potassium Page Three
References
A list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.
Related Resources
- Anatomy of the Atom
Answers many questions regarding the structure of atoms. - Molarity, Molality and Normality
Introduces stoichiometry and explains the differences between molarity, molality and normality. - Molar Mass Calculations and Javascript Calculator
Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. - Chemical Database
This database focuses on the most common chemical compounds used in the home and industry.
Citing this page
If you need to cite this page, you can copy this text:
Kenneth Barbalace. Periodic Table of Elements - Potassium - K. EnvironmentalChemistry.com. 1995 - 2021. Accessed on-line: 4/24/2021
https://EnvironmentalChemistry.com/yogi/periodic/K.html
.
https://EnvironmentalChemistry.com/yogi/periodic/K.html
.
Linking to this page
If you would like to link to this page from your website, blog, etc., copy and paste this link code (in red) and modify it to suit your needs: How to add gmail contacts to iphone 11.
<a href='https://EnvironmentalChemistry.com/yogi/periodic/K.html'>echo Periodic Table of Elements: Potassium - K (EnvironmentalChemistry.com)</a>- Comprehensive information for the element Potassium - K is provided by this page including scores of properties, element names in many languages, most known nuclides and technical terms are linked to their definitions.
.
.
NOTICE: While linking to articles is encouraged, OUR ARTICLES MAY NOT BE COPIED TO OR REPUBLISHED ON ANOTHER WEBSITE UNDER ANY CIRCUMSTANCES.
PLEASE, if you like an article we published simply link to it on our website do not republish it.
